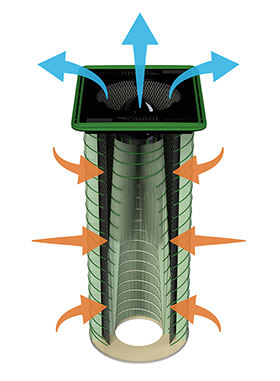
The dust collector designed specifically for solid dose pharmaceutical and containment applications
The Gold Series (GS) Camtain dust collector for pharmaceutical applications can be used for tablet presses, coating, fluid bed drying, spray drying, blending, granulation and general room ventilation.
It is perfect for high-efficiency filtration where recovery of the product is not required. The GS Camtain also support traditional dust collection for nuisance dusts and fumes that do not require full isolation and containment.
Camfil has a team available to support the pharmaceutical market. Qualified engineers will work with you to properly design and install the right Gold Series Camtain dust collectors for your specific applications.
In addition to the safe-change containment systems above, the GS Camtain has special options just for pharmaceutical applications including:
- Pharmaceutical Upgrade Package
- Heavy-duty construction and stiffeners rate housing to 0.44785 bar
- Deflectors to eliminate all internal horizontal ledges
- Closed cell silicone gaskets and caulk at all bolted connections
- HEPA filter on the pressure taps for the pressure differential gauge
- Explosion Vents
- Burst detector
- Vertical plenum
- Up to 1016 mm diameter vent duct
- Weather hood
- Chemical Suppression
- Chemical Isolation
The Gold Series Camtain is now specified by most major pharmaceutical companies.
- BOSCH Manesty
- GlaxoSmithKline
- AstraZeneca
- Pfizer
- Abbott
- Biovail
- Bristol-Myers Squibb
- Eli Lilly and Company
- Hikma
- Baxter
- Perrigo
- Novartis
- Tyco Mallinckrodt (Covidien)
- Upsher-Smith
- Merck
- Warner Chilcott
- J & J Ortho-McNeil
- J & J Janssen
- GSK
- Sandoz - Eon Labs
- Aurobindo/Aurolife
- Bayer
- Genzyme
- West Ward (BIRI)
- Amneal
- Thomas Engineering
- GEA
- Glatt
- Vector
- O’Hara Technologies
- L.B. Bohle
- Jacobs
- Fluor
- CE&IC
- CRB
- IPS
- Stantec

Key features of the Camtain
Safe-change containment systems are available for both the filter cartridges and discharge system underneath the collector. The cartridge change utilizes the safe-change filter replacement method while the discharge uses continuous liner technology.
Filter cartridges are sealed via an internal cam bar action, allowing convenient changeout through the safe-change filter replacement system.
High-entry, cross flow inlet eliminates upward velocities that can hold fine particulates in the filter cartridges, reducing the re-entrainment of the particulate matter.
Vertically arranged filter cartridges allow captured particles to release directly to the hopper instead of onto other filter cartridges (which happens when using horizontal-oriented cartridges.)
High-efficiency filters up to MERV 15 per ASHRAE 52.2:2007
Discover more – connect with our team
Optimized design advantages

Surrogate tested
The Gold Series Camtain dust collector for pharmaceutical applications has been independently surrogate tested for validated performance verification. A major pharmaceutical company’s surrogate testing protocol was followed by an independently contracted, AIHA-accredited laboratory (Bureau Veritas) test.
Using 100% milled lactose as the surrogate, we collected over 75 area, personal and swab samples for both the safe-change filter replacement and the continuous liner discharge. The GS Camtain can handle risk-based category 3, 4 and 5 compounds with occupational exposure limits less than 1.0 mcg/m3 for an 8-hour time-weighted average.
Full test report data is available upon request. You can download a summary of the surrogate test report now.

Worker health/containment
It's critically important to understand the toxicological properties of the pharmaceutical dusts being collected. This is necessary to determine the occupational exposure limit (OEL) and perform a risk-based exposure evaluation to determine the methods for proper control.
In most cases, some level of isolation and containment is required for dusts being captured in a non-production areas. This is because pharmaceutical dust is extremely potent and can not be released into the surrounding environment. Camfil offers safe-change containment systems for both the filter cartridges and discharge system. The cartridge change utilizes a safe-change filter replacement method while the discharge uses continuous liner technology.
In most cases, Camfil APC recommends a HEPA secondary polishing system. A HEPA backup systems enables the filtered air to be recirculated air back into the HVAC system. This can significantly reduce energy costs while providing the necessary level of filtration for discharge air required by the EPA.

Explosion protection and compliance
A critical consideration when designing dust collectors for pharmaceutical applications is protecting workers from deflagration and explosion risks. Control measures such as explosion venting, chemical suppression and isolation systems may be required depending on the physical characteristics of the dust relating to Kst, MIE and the location of the collector.
When explosion vents are required, they must be vented to the outside by either placing the collector outdoors or ducting the vent exhaust a specified distance through the building structure. To prevent outdoor or off-property exposure in the event of a dust explosion of potent compounds, either chemical suppression or containment designs for explosion and isolation valves are preferable to venting. The reason why is because these two protection options do not release materials to the environment.
Camfil APC recommends that an independent PE specifies what explosion protection is required for a given material as it relates to standards in NFPA, ATEX and the major insurance carriers.


 Americas
Americas